The Activation Energy Is Best Described by
This is the starting energy before we go on the reaction. The experimental data were best described by a first-order autocatalytic equation with activation energies equal to 178 and 175 kJmole for the two steps of the autocatalytic reaction.
Activation Energy Article Khan Academy
The temperature at which the reaction proceeds most quickly.

. C the energy of motion. The energy level of the products. Particles need to collide with each other with minimum amount of energry.
If less energy is available a chemical reaction is unable to proceed. The catalysts needed to raise a reactions rate. Activation energy is the minimum amount of energy required by chemical reactants to undergo a chemical reaction.
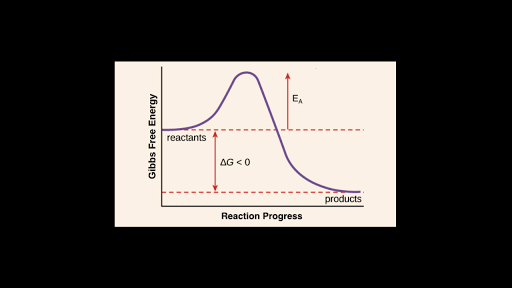
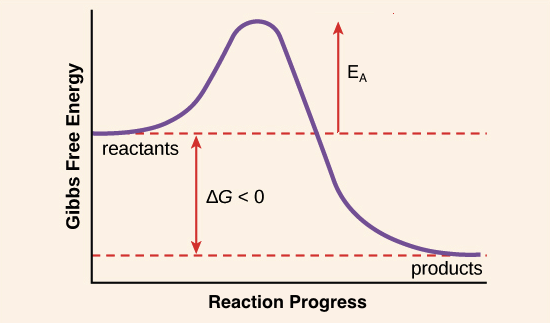
The diagram in figure 13 shows the amount of energy the reactions starts and how much energy is being released or absorbed depending on the reaction that is occurring. The answer is B as activation energy is the least possible amount of energy minimum which is required to start a reaction or the amount of energy available in a. 1energy input needed to break bonds of reactants.
Science Chemistry QA Library Which statement below best describes the relationship between activation energy and the rate constant. So I think its C. Rank the grades of coal by their relative desirabilities starting with mos desirable at the top.
A numerical description of the amount of energy needed by colliding reactant molecules in order to form. The energy required for a reaction to proceed by breaking bonds d. The difference in energy between products and reactants d.
A type of endergonic reaction c. The combined energy of all the reactants b. The greater the activation energy the lower the reaction rate.
Which of the following best describes the activation energy of a chemical reaction. D the energy required to separate ions in a crystalline solid. The amount of energy needed to activate a reaction or for it to occur.
White phosphorus has a higher activation energy than red phosphorus. Activation energy is also defined as the least possible energy required to initiate a chemical. 4Products have higher chemical potential energy than reactants.
The difference in energy between reactants and the maximum energy. The difference in free energy between. Group of answer choices a The amount of energy lost in an exothermic reaction.
The energy required for a reaction to proceed by breaking bonds. Because activation energy is the amount of energy required for a chemical reaction to take place. How can the chemical potential energy in an endothermic reaction best be described.
The word activation energy in chemistry is known to be the little or minimum amount of energy that is needed by one to be able to ignite or activate atoms or molecules. The difference in free energy between the products and the transition state. The energy level of the reactants.
And if the energy of the colliding molecules is less than the activation energy no reaction will also occur. B The amount of energy gained in an endothermic reaction c All of the above d The amount of energy required to start a reaction. The potential energy stored in the bonds of reactants and products.
This is the starting energy before we go on the reaction. Because activation energy is the amount of energy required for a chemical reaction to take place. White phosphorus has a lower activation energy than red phosphorus.
The Activation Energy can best be described as. O As the activation energy increases the rate constant decreases O As the activation energy increases the rate constant decreases O The activation energy and rate constant are both dependent upon temperature O As the activation energy. Activation energy simply means the minimum amount of energy needed for the particles to have a successful reaction when they collide.
Activation energy can best be described as. Activation energy is the energy absorbed before it can start a chemical reaction. Products have higher chemical potential energy than reactants.
Answer 1 of 2. B the minimum kinetic energy that particles must possess for a chemical reaction to occur. Energy difference between the reactants and the products.
The molecules or atoms are activated to a condition where they can be said to undergo chemical transformation or a kind of physical transport. This will give you a value of energy measured in joules If you use the r. White phosphorus has a lower activation energy than red phosphorus.
How do temperature and activation energy relate to. No variation in activation energy was observed between the different NC samples whatever their nitrogen content and vegetal origin or the solvent used to cast films. Which best describes the energy of activation.
3White phosphorus has a lower activation energy than red phosphorus. Activation energy is best described as ___ the energy required to initiate a chemical reaction. The difference in free energy between the reactants and products of the reaction D.
Reducing the activation energy can increase the rate of a reaction. The activation energy in the Arrhenius equation can best be described as asked Jun 26 2017 in Chemistry by maju88 a. If you need to kick a ball over a wall you can calculate the potential energy that the ball would have at the top of the wall as mgh mass of ball times acceleration of gravity times height of wall.
The amount of energy required for a reaction to occur c. 2energy stored in chemical bonds. If there is no activation energy no reaction will occur.
4How can the chemical potential energy in an endothermic reaction best be described1 point Reactants have higher chemical potential energy than products. Reducing the activation energy would mean that less energy is required to process a reaction whereas increasing activation energy would require more energy to process a reaction. Activation energy is a concept used in chemistry that was introduced by the scientist from Sweden named Svante Arrhenius in 1889.
So I think its C. Which of the following best describes the activation energy of a reaction. Energy difference between the reactants and the activated complex.
A type of exergonic reaction b. The maximum energy level of the reaction. Activation energy can be described as the a.
The difference in energy between reactants and products. Energy of the activated complex. Activation energy is the amount of energy required to initiate a chemical reaction.
There are two basic factors necessary for a reaction to just take place. The activation energy can be determined by reaction rate constants at different temperatures by the equation.
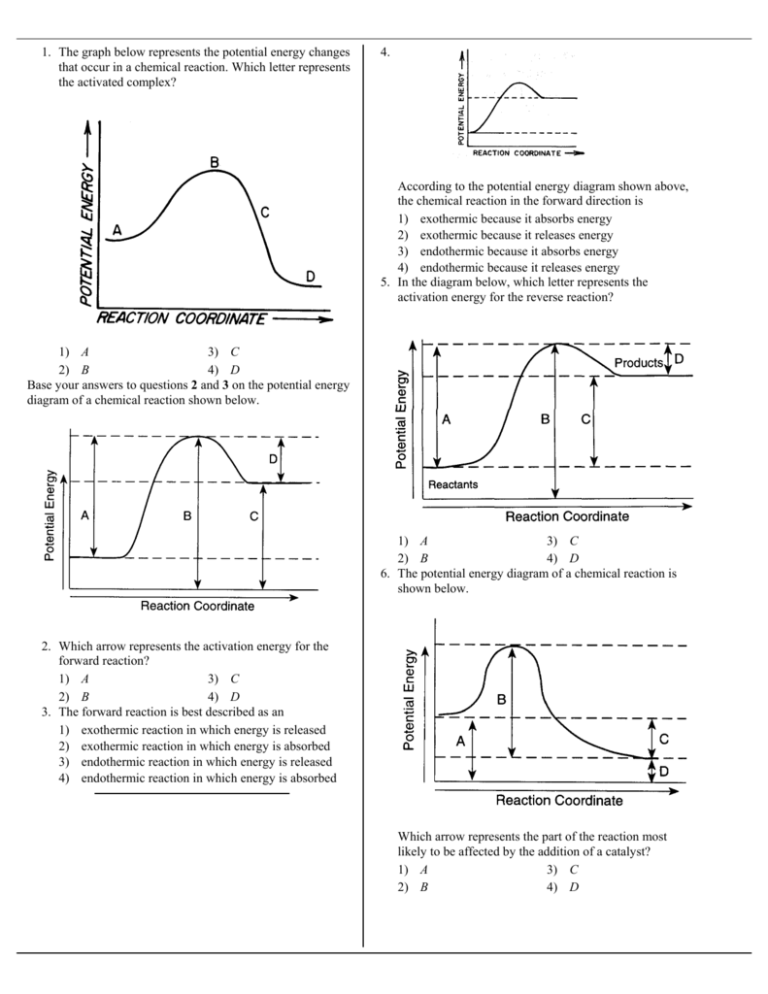
1 The Graph Below Represents The Potential Energy

Kinetics Why Is Activation Energy Drawn In A Potential Energy Diagram In Reactions Chemistry Stack Exchange
Comments
Post a Comment